肥胖被认为是慢性疾病的主要危险因素,如冠心病和高血压、2型糖尿病和某些癌症。富含脂肪的饮食会导致小鼠肥胖。在小鼠中,已经发现饮食中的脂肪水平与体重或脂肪增加呈正相关。通常使用占总能量摄入30-78%的高脂肪饮食——可以选择在动物的饮食中添加特定的脂肪,或者使用各种富含脂肪和糖的超市/自助餐饮食品。
Dulaglutide对DIO小鼠模型的抗肥胖作用

Dulalgutide在高脂饮食诱导的肥胖(DIO)小鼠模型中的抗肥胖作用C57BL/6小鼠给予高脂饲料(60 kcal% Fat)喂养12周诱导肥胖。分组后每周2次皮下注射度拉鲁肽(内部合成),共4周。末次给药后次日进行葡萄糖耐量实验(n=8 /组)。
Celastrol对高脂饮食诱导肥胖(DIO)小鼠模型的抗肥胖作用
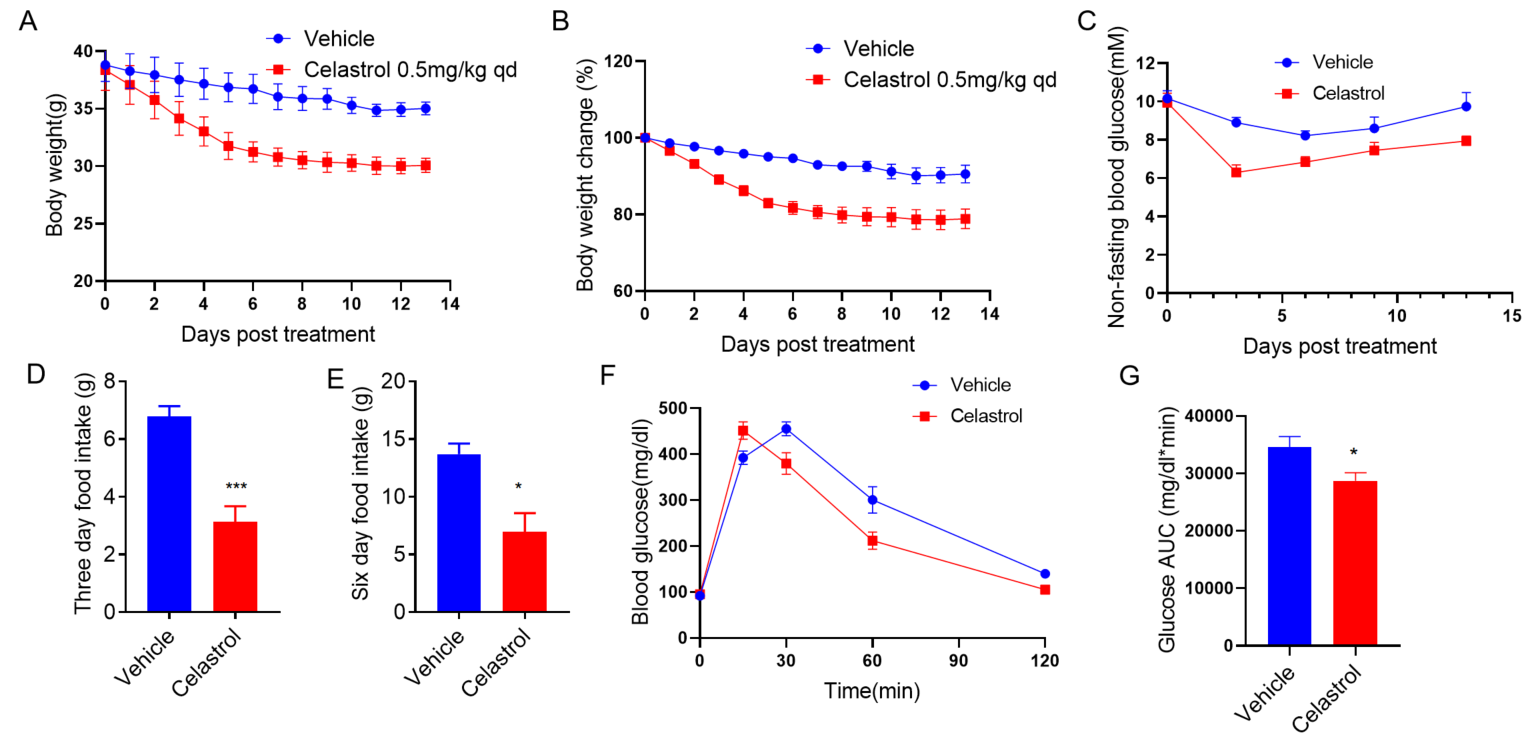
C57BL/6小鼠给予高脂饲料(60 kcal% Fat)喂养12周诱导肥胖。然后腹腔注射Vehicle或Celastrol(0.5mg/kg),每天1次,连续14天,体重(A、B)、血糖(C)、进食量(D、E)和葡萄糖耐量试验(F、G)结果如上所示(n=5 /组)。
糖尿病是目前威胁人类健康的重要疾病。近20年来,我国糖尿病患病率迅速上升,目前患病人数已接近9000万。ob/ob小鼠是目前研究人类2型糖尿病的最佳动物模型之一。模型小鼠的生物学特性和生理生化指标是各种实验中共同且重要的参考指标。
Dulaglutide对B-ob/ob小鼠的抗肥胖和降血糖作用
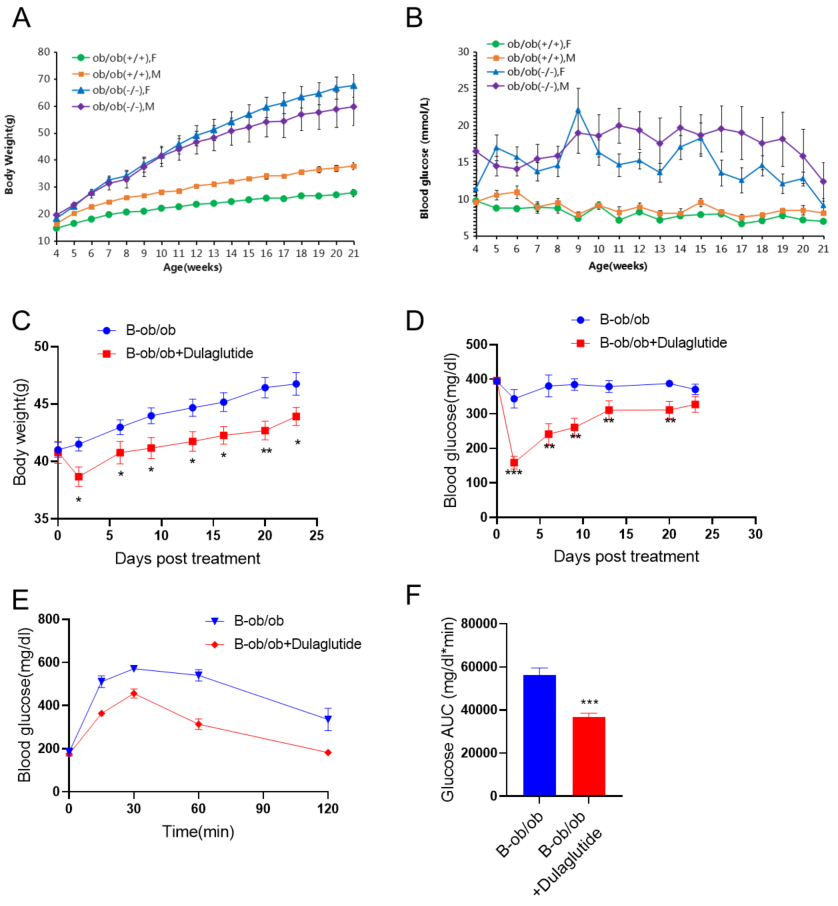
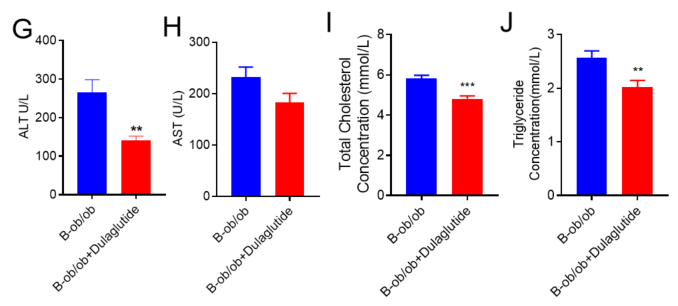
(A-B) 4 ~ 21周龄B-ob/ob小鼠体重和血糖变化。(C-F) 9周龄B-ob/ob小鼠分组后每周2次给予1mg/kg度拉鲁肽(内部合成)灌胃4周。在指定时间点记录体重和血糖。末次给药后次日进行葡萄糖耐量实验(n=8 /组)。(G-J)血液生化检测显示度拉鲁肽对B-ob/ob小鼠的影响(n=8只/组).
Celastrol对B-ob/ob小鼠的抗肥胖和降血糖作用
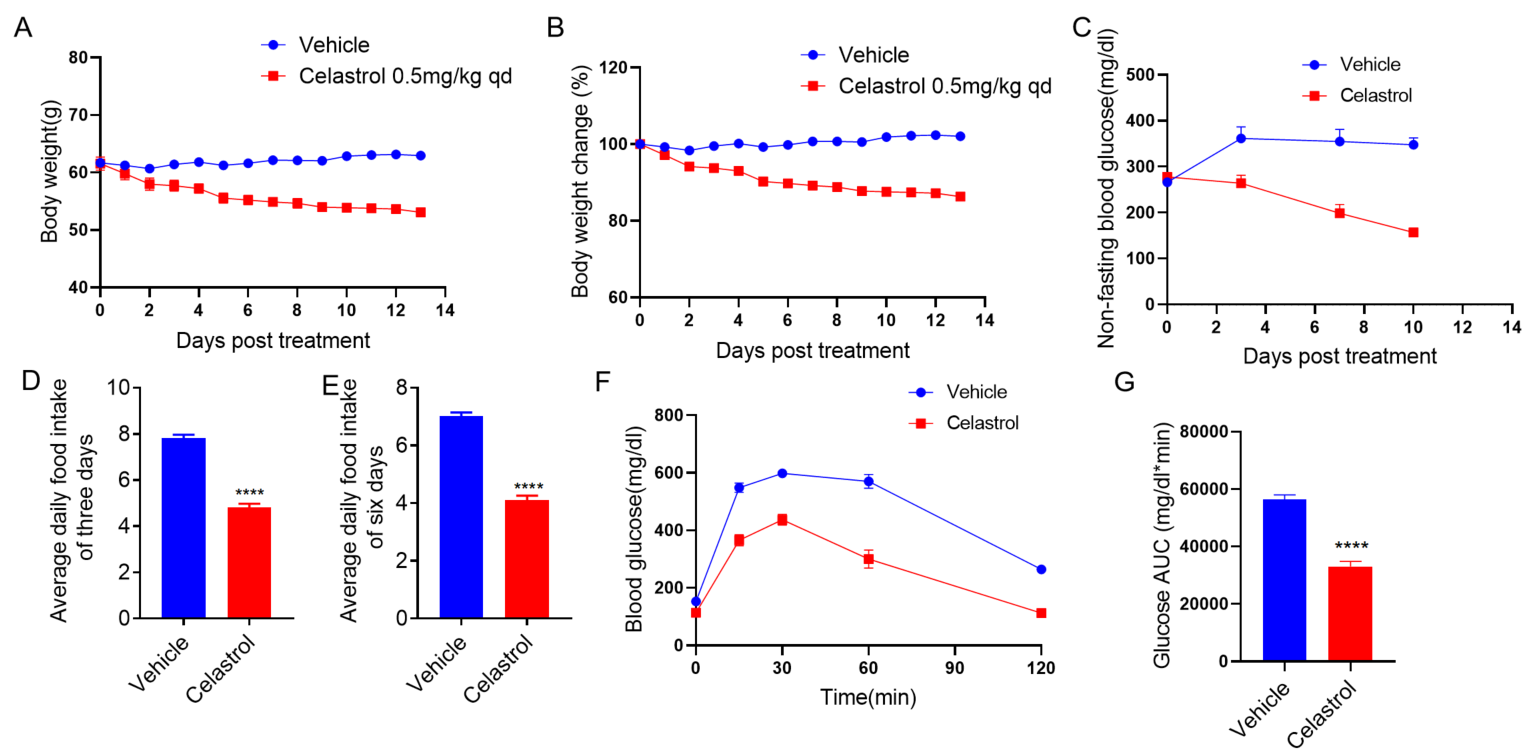
雄性B-ob/ob小鼠腹腔注射Vehicle或Celastrol (0.5mg/kg),每天1次,连续14天,体重(A、B)、血糖(C)、进食量(D、E)和葡萄糖耐量试验(F、G)结果如上所示。(n=8 /组)。
链脲佐菌素(STZ)是一种可破坏胰岛β细胞的抗生素,在实验中广泛用于制造1型糖尿病(T1DM)模型。STZ是一种含亚硝基的化合物,可通过诱导一氧化氮(NO)的合成,特异性破坏胰岛B细胞,增加胰岛B细胞的氧化侵袭。目前普遍认为,小剂量多次注射STZ可诱导实验动物发生1型糖尿病。其机制可能是注射小剂量STZ后,少量胰岛细胞被破坏,死亡的胰岛细胞被巨噬细胞释放致敏蛋白作为抗原。吞噬作用,产生th1刺激因子(IL-12),诱导IL-2和IFN-γ分泌增加,在局部胰岛形成炎性细胞浸润,释放IL-1β、TNF-α、IFN-γ等细胞因子和氧自由基杀死少量细胞。死亡细胞将自身作为抗原,经APC细胞处理后释放细胞因子,放大细胞损伤效应,最终导致1型糖尿病.
STZ诱导糖尿病小鼠模型的建立
GCGR抗体对STZ诱导的B-hGCGR小鼠的降糖作用
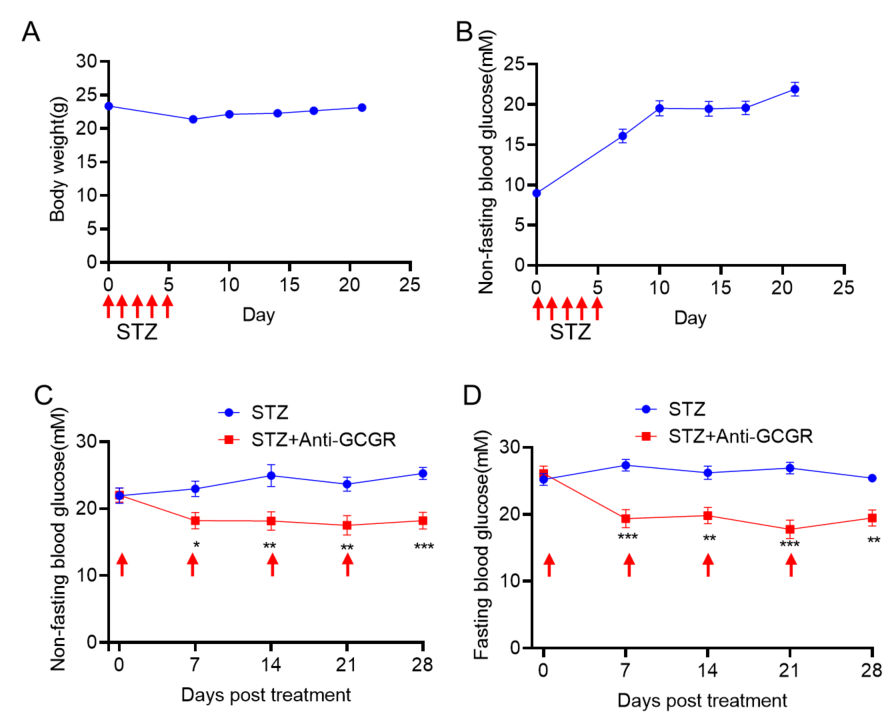
GCGR抗体对STZ诱导的B-hGCGR小鼠的降糖作用。(A-B) STZ (50mg/kg)诱导6周龄B-hGCGR小鼠体重和血糖变化,(C-D) STZ诱导后3周,GCGR抗体(10mg/kg,内部合成)每周给药1次,测定非空腹血糖(C)和空腹血糖(D).
高脂饮食诱导肥胖(DIO)小鼠模型

自发性糖尿病模型(B-ob/ob小鼠)

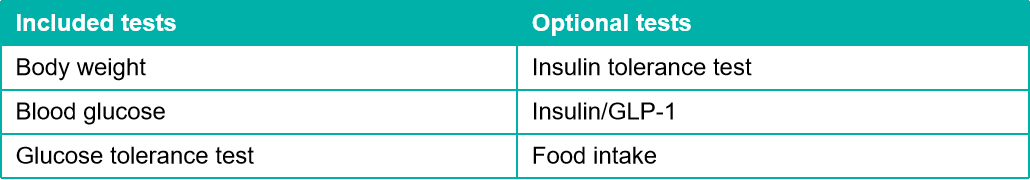
STZ诱导糖尿病模型
 苏公网安备:32068402320845号
网站建设:北京分形科技
苏公网安备:32068402320845号
网站建设:北京分形科技






 010-56967680
010-56967680 info@bbctg.com.cn
info@bbctg.com.cn